A Strange Mating System
Spurred by an incidental observation of two color-banded males sequentially feeding young at a Stratton Mountain nest, we delved further by placing video cameras at nearly one hundred nests to identify feeders. (Watch a female feed her nestlings). Then we analyzed mitochondrial DNA of nestlings and adults.
-

© Kent McFarland
To our and many others’ great surprise, we found that two to as many as four males attended 75% of the nests, but never more than a single female did so. Coincidentally, we learned that males do not hold traditional territories but wander widely over home ranges of up to 20 hectares (50 acres), each bird broadly overlapping its movements with those of up to seven other males. Females, in contrast, occupy and defend much smaller, non-overlapping territories.
The plot thickened when we analyzed paternity, which was highly mixed in 70% of nests. In short, both male and female Bicknell’s Thrushes mate with multiple partners, some males feed multiple broods concurrently, some males feed broods in which they have no paternity, and females on higher quality territories (as defined by arthropod prey biomass) fledge more chicks and have fewer males feeding them.
This complex breeding system, termed “female-defense polygynandry,” is known in only one other North American songbird: Smith’s Longspur.
Scientific Publications
Goetz, J.E.., K. P. McFarland and C.C. Rimmer. 2003. Multiple paternity and multiple male feeders in Bicknell’s Thrush (Catharus bicknelli). Auk 120: 1044-1053. (Abstract)
Strong, A.M., C.C. Rimmer, and K.P. McFarland. 2004. Effect of prey biomass on reproductive success and mating strategy of Bicknell’s Thrush (Catharus bicknelli), a polygynandrous songbird. Auk 121:446-451. (Abstract)
Balsam Fir: A Keystone Species
We annually monitored balsam fir reproduction, bird densities and reproductive success, and red squirrel densities, and discovered a biennial cycle.
-

© Alden Wicker
High cone production occurred biennially in late summer and fall of even-numbered years, and was followed by immigration and breeding by White-winged Crossbills, Pine Siskins and red squirrels. Open cup-nesting birds experienced extremely low rates of nest success in summers following heavy cone mast, probably due to nest depredation by red squirrels and other small mammals, and high success following mast failures. Analysis of demographic variables indicates that our study populations are sinks in odd years, sources in even years, and barely break-even overall.
Read More
A Good Year for Fir Cones, by Kent McFarland, North Woodlands Magazine – Outside Story 2003.
Songbirds and Ski Area Development
Overall, few significant differences existed for various population and reproductive parameters between areas developed for ski areas and natural forests on each mountain. We found no evidence that nest predation rates differed between ski area and natural forest plots, or that nests in either plot type were more likely to be depredated.
Despite higher nest densities near ski trail edges, it appears that edge effects do not exert an important influence on rates of nest predation for Bicknell’s Thrush, and that the ‘ecological trap’ hypothesis does not apply to this species in existing ski areas. There was no evidence that female brooding behavior, male feeding behavior, or movements of adults differed between ski areas and natural forests, although our study design did not specifically address these issues. Finally, we found no significant differences in adult survivorship, nest success, or breeding productivity of Bicknell’s Thrushes between ski areas and natural forests, although a power analysis suggested that survival probabilities for adult males were somewhat lower in ski-slope edge habitats.
Findings from radio telemetry of adult thrushes indicated that trail crossings wider than 50 meters were avoided, and that small or narrow habitat islands were rarely used. As a general rule, ski trails greater than 35 to 40 meters in width appear likely to restrict thrush movements. Breeding thrushes were concentrated in habitat blocks consisting of fairly large, closely adjoining islands intersected by narrow, winding ski trails; these were often separated by large open areas. Habitat configurations that featured alternating narrow, linear slopes and islands tended not to support clusters of Bicknell’s Thrush home ranges.
This suggests that trail design should maximize habitat contiguity and minimize fragmentation to promote suitable features for Bicknell’s Thrush. Fewer, larger islands separated by narrow, non-linear trails are preferable to larger numbers of small islands or alternating bands of linear trails and islands.
Scientific Publications
Rimmer, C.C., Kent P. McFarland, J. Daniel Lambert, and Rosalind B. Renfrew. 2004. Evaluating the use of Vermont ski areas by Bicknell’s Thrush: applications for Whiteface Mountain, New York. Unpublished report. (PDF)
Strong, A. M., C. C. Rimmer, K.P. McFarland and K. Hagan. 2002. Effects of mountain resorts on wildlife. Vermont Law Review 26(3): 689-716. (PDF)
Bicknell’s Thrush Habitat and Climate Change
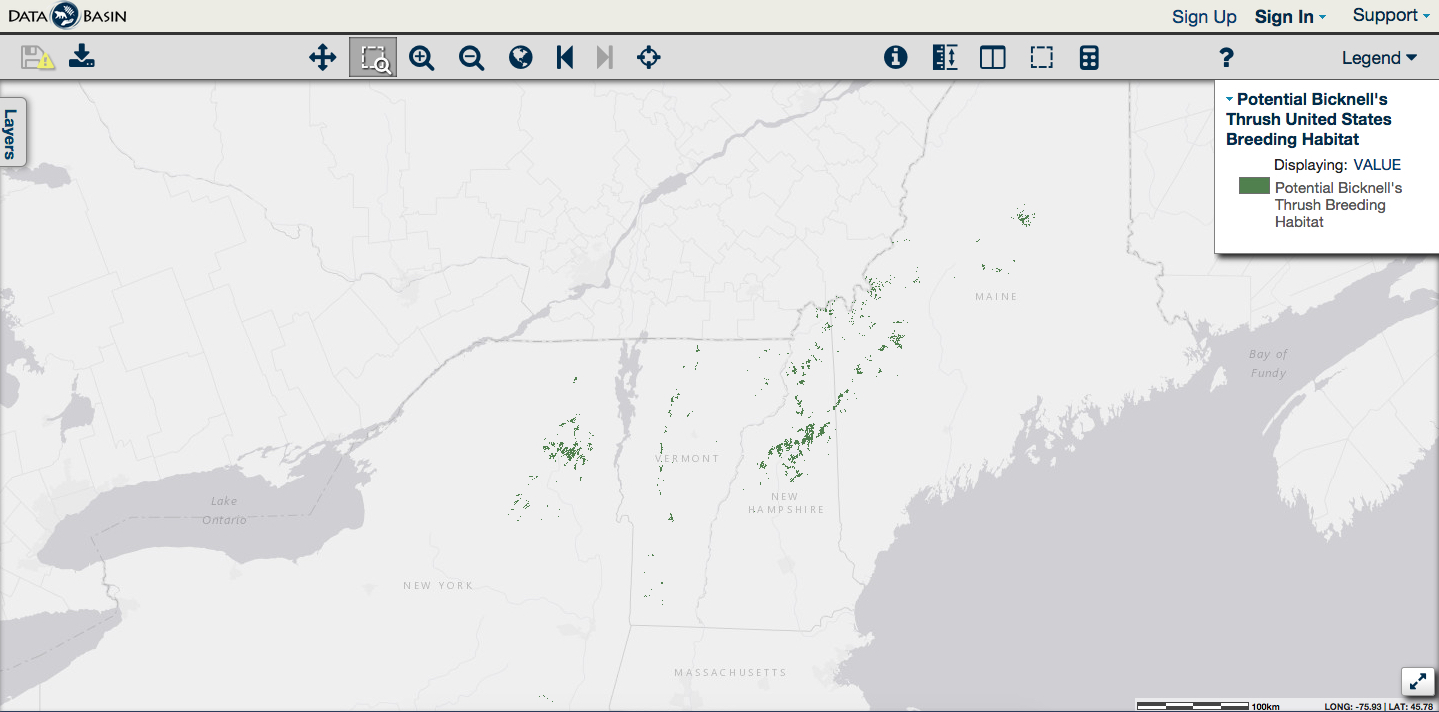
Click on the map to visit DataBasin, where you can use an interactive map of potential Bicknell’s Thrush habitat and download data for your GIS.
For the first time, we can reliably predict where Bicknell’s Thrush breeding habitat is located in the northeastern United States. This information is in great demand from natural resource agencies, conservation groups, and those who own land in the high country. The map identifies potential habitat by integrating three layers of geographic information: forest type, elevation, and latitude. It shows nearly 340,000 acres of red spruce and balsam fir forest in mountainous regions of New York, Vermont, New Hampshire, and Maine. In a test of the map’s accuracy, it proved to be 85% to 98% correct in predicting the presence or absence of Bicknell’s Thrush along 1-km survey routes.
This map helps to identify opportunities to conserve critical habitat and it provides a basis for evaluating plans to develop mountain areas. It also allows us to examine what might happen to this habitat in the future under different climate change scenarios.
Global warming is poised to substantially change the climate in the Northeast if heat-trapping emissions are not curtailed, and the extent and impacts of the change depend on the choices that governments, businesses and citizens make today. By the end of this century, for instance, summers in Vermont could feel like those in Tennessee if emissions continue unabated. But if emissions are reduced, summers in Vermont could resemble those of West Virginia. So concludes the study released by the Northeast Climate Impacts Assessment, a collaboration between the Union of Concerned Scientists and a team of independent scientists from universities across the Northeast and the nation, including VCE biologists.
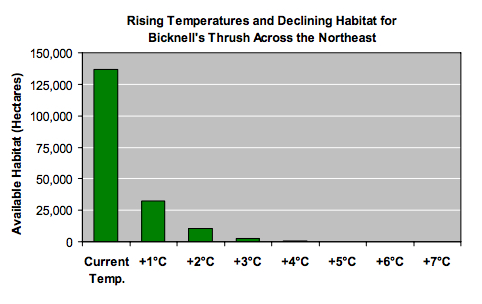
Under the rising mean summer temperatures examined by the VCE biologists, the spruce-fir zone literally loses ground. Warming climate causes the lower boundary of this zone to gradually retreat up the mountain. The spruce-fir forest and its suite of plant and animal communities are therefore limited to progressively higher, smaller, and more isolated patches.
Our research shows that the most vulnerable species may be those that, like the Bicknell’s thrush, depend on high elevation spruce-fir habitat. Under either low or high emissions scenarios, great losses in suitable habitat are expected for this rare songbird as a result of climate changes projected this century. Only under the lower-emissions scenario is this range-restricted species projected to retain more than 10 percent of its U.S. habitat. Mountain-breeding populations of spruce grouse, three-toed woodpecker, black-backed woodpecker, gray jay, yellowbellied flycatcher, boreal chickadee, and blackpoll warbler are expected to be similarly affected.
- A summer temperature increase of roughly 4°F, projected by mid-century under either emissions scenario, may be enough to eliminate all breeding sites for the Bicknell’s thrush at the southern edge of its range in the Catskill Mountains of New York and most of Vermont.
- With summer warming of 9°F, projected under the higher-emissions scenario by late this century, only small patches of suitable habitat for the Bicknell’s thrush may remain in New Hampshire’s Presidential Range and on Mount Katahdin in Maine.
- If summers warm by 11°F, also possible by late in the century under the higher-emissions scenario, suitable habitat for the Bicknell’s thrush is expected to disappear from the Northeast.
- In the Berkshires of Massachusetts and the Allegheny Plateau in Pennsylvania, encroachment of hardwoods into mountaintop spruce/fir forests (projected as late-century warming reduces spruce/fir habitat) would threaten the remaining small populations of species such as the blackpoll warbler and yellow-bellied flycatcher. Further north, these species are less vulnerable because they occur in both high- and low-elevation spruce/fir forests.
Mercury in the Mountains
Mercury is a pollutant that is cause for concern on local, regional, and global scales. Because mercury released into the atmosphere can circle the globe before being deposited, habitats located far from point sources can still be of major concern for wildlife health. The mountains in northeastern North America are one of those places.
From 2001 to 2005, our scientists were part of a landmark mercury research partnership to compile mercury data from across the northeastern United States and Canada. As a result of that effort, 21 scientific papers were published in a special issue of the scientific journal Ecotoxicology and summarized in a report by Biodiversity Research Institute, Mercury Connections: The Extent and Effects of Mercury Pollution in Northeastern North America.
Previously, most research had focused on mercury contamination in fish and fish-eating birds in aquatic environments. Our data, reported in the Ecotoxicology volume, revealed that Bicknell’s Thrush and other songbirds were accumulating mercury, and nearly all of it in the toxic methyl form. This finding has implications for the way scientists and policymakers view the nature and extent of mercury in northeastern North America.
Offering novel insights into mercury’s threat to wildlife, our scientists for the first time revealed how this insidious toxin moves and concentrates across the food chain in forest ecosystems. VCE biologists Chris Rimmer, Kent McFarland, and Steve Faccio, working as mercury detectives on Vermont’s Stratton Mountain, tracked the toxin at discrete levels in the mountain’s food web: leaf litter, foliage, foliage-eating insects, omnivorous arthropods (insects and snails, for example), a terrestrial salamander (Red-backed Salamander), an insectivorous songbird (Bicknell’s Thrush) and two raptors (Sharp-shinned Hawk and Northern Saw-whet Owl). Their goal was to examine mercury concentrations and variability in organisms at different trophic levels during summer.
As might be expected, Sharp-shinned Hawk and Northern Saw-whet Owl, at the top of the montane forest food web, showed the highest mercury levels in their blood. Although leaf litter might be considered the base of the food web, this zone showed relatively high levels of mercury as well, higher than any other biotic compartment except Sharp-shinned Hawk. One explanation for this is that mercury can accumulate and persist over time in the leaf litter, leaching out of the fallen foliage during decomposition.
In live plant foliage, balsam fir needles showed greater concentrations of mercury than the three deciduous tree species VCE sampled: paper birch, mountain ash, and pin cherry. Fir needles grow and remain on a tree for up to three years, accumulating mercury each year, compared to only one-year’s growth in deciduous species. Even so, the three deciduous species showed increasing mercury levels throughout the season, at a rate higher than that observed in balsam fir needles
Moving up the food web to insects and other arthropods, mercury concentrations ranged widely but were lowest in herbivorous insects (such as grasshoppers, leafhoppers, and moth caterpillars), and higher in the predatory insects (such as scorpionflies and lacewings). Finally, among all arthropods sampled, carnivorous spiders and harvestmen showed the highest mercury concentrations, an expected result among these longer-lived predators of other insects.
 Red-backed Salamander on 380 million-year-old schist on Mt. Mansfield, Vermont.
Red-backed Salamander on 380 million-year-old schist on Mt. Mansfield, Vermont.
Red-backed Salamanders showed relatively high concentrations of mercury, suggesting that they feed at higher trophic levels within the invertebrate community or that they prefer prey species that themselves accumulate relatively high amounts of mercury. As it turns out, Red-backed Salamanders live and forage in moist soils, often near stream edges, where methylmercury concentrations may be relatively high.
Perhaps the study’s most compelling discovery involved Bicknell’s Thrush. Mercury levels in this signature montane songbird spiked early in the season and decreased over the course of the summer. A likely explanation is that when Bicknell’s Thrush returns to breeding sites in May, spiders, harvestmen, predatory beetles, and other prey species relatively high in mercury are more numerous and probably constitute a significant portion of the songbird’s diet. Snails, also relatively high in mercury, may be consumed by female thrushes to mobilize additional calcium for egg production. But as new coniferous and deciduous foliage emerges during June, Bicknell’s Thrush almost certainly shifts its diet to newly abundant populations of leaf-eating insects with lower mercury levels. This seasonal dietary shift likely explains the decline in thrush blood mercury concentrations between early and late summer.
Results of VCE’s investigation provide critical evidence that mercury bioaccumulates and biomagnifies in the montane forest biotic community. Bicknell’s Thrush, already receiving ample attention from VCE, may serve as a valuable bioindicator for the continued monitoring of mercury contamination in terrestrial food webs. VCE’s previous work has already demonstrated that high-elevation coniferous forests are particularly vulnerable to climatic warming. The cumulative impacts of climate change, mercury toxicity and other stressors on these geographically restricted montane habitats could profoundly threaten their unique assemblage of flora and fauna, as well as aesthetic and recreational opportunities for millions of people in the northeastern United States.
Read more about it in one of VCE’s earliest publications, Research Notes.
Scientific Publications
- Townsend, J.T., C.T. Driscoll, C.C. Rimmer, and K.P. McFarland. 2014. Avian, salamander, and forest floor mercury concentrations increase with elevation in a terrestrial ecosystem. Environmental Toxicology and Chemistry 33:208-215. (Abstract)
- Townsend, J.M., C.C. Rimmer, C.T. Driscoll, K.P. McFarland, and E.E. Iñigo-Elias. 2013. Mercury concentrations in tropical resident and migrant songbirds on Hispaniola. Ecotoxicology Volume 22 (1): 86-93. DOI 10.1007/s10646-012-1005-1. (Abstract)
- Rimmer, C.C., E.K. Miller, K.P. McFarland, R.J. Taylor, and S.D. Faccio. 2009. Mercury bioaccumulation and trophic transfer in the terrestrial food web of a montane forest. Ecotoxicology 19(4): 697-709. (Abstract)
- Rimmer, C.C., K. P. McFarland, D. C. Evers, E. K. Miller, Y. Aubry, D. Busby, and R. J. Taylor. 2005. Mercury levels in Bicknell’s thrush and other insectivorous passerine birds in montane forests of the northeastern United States and Canada. Ecotoxicology 14:223-240. (Abstract)
Scientific Publications
Deckel, S.C., Narango, D.L., DeLuca, W., King, D.I., Gerson, A. In Press. Stable Isotope Analysis and DNA Metabarcoding Reveals Elevational Shifts in Diet of a Montane Breeding Bird. Journal of Field Ornithology.
Lewis, W.B., R.J. Cooper, R.B. Chandler, R.W. Chitwood, M.H. Cline, M.T. Hallworth,, J.L. Hatt, J. Hepinstall‐Cymerman, S.A. Kaiser, N.L. Rodenhouse, and T.S. Sillett. 2023. Climate‐mediated population dynamics of a migratory songbird differ between the trailing edge and range core. Ecological Monographs, 93(1), p.e1559.
Hagelin, J.C., M.T. Hallworth, C.P. Barger, J.A. Johnson, K.A. DuBour, G.W. Pendelton, L.H. DeCicco, L.A. McDuffie, S.M. Matsuoka, M.A. Snively, P.P. Marra, and A. Taylor. 2021. Revealing migratory path, important stopovers and non-breeding areas of a boreal songbird in steep decline. Animal Migration 8:168–191.
Hill, J.M., J.D. Lloyd, K.P. McFarland, and C.C. Rimmer. 2019. Apparent survival of a range-restricted montane forest bird species is influenced by weather throughout the annual cycle. Avian Conservation and Ecology 14:16. doi.org/10.5751/ACE-01462-140216
Hill, J.M. and J.D. Lloyd. 2017. A fine-scale U.S. population estimate of a montane spruce–fir bird species of conservation concern. Ecosphere 8(8):e01921. doi.org/10.1002/ecs2.1921
DeLuca, W.V., B.K. Woodworth, C.C. Rimmer, P.P. Marra, P.D. Taylor, K.P. McFarland, S.A. MacKenzie, and D.R. Norris. 2015. Transoceanic migration by a 12 g songbird. Biology Letters 11:20141045.
Rimmer, C.C. and K.P. McFarland. 2013. Bicknell’s Thrush: a twenty-year retrospective on the Northeast’s most vulnerable songbird. Bird Observer 41:9-16.
Studds, C. E., McFarland, K. P., Aubry, Y., Rimmer, C. C., Hobson, K. A., Marra, P. P. and Wassenaar, L. I. 2012. Stable-hydrogen isotope measures of natal dispersal reflect observed population declines in a threatened migratory songbird. Diversity and Distributions 18: 919–930. (Abstract)
Rimmer, C.C., E.K. Miller, K.P. McFarland, R.J. Taylor, and S.D. Faccio. 2009. Mercury bioaccumulation and trophic transfer in the terrestrial food web of a montane forest. Ecotoxicology 19(4): 697-709. (Abstract)
Frey, S.J.K., C.C. Rimmer, K.P. McFarland, and S. Menu. 2008. Identification and sex determination of Bicknell’s Thrushes using Morphometric Data. J. of Field Ornithology 79: 408-420. (Abstract)
Rimmer, C.C., K. P. McFarland, D. C. Evers, E. K. Miller, Y. Aubry, D. Busby, and R. J. Taylor. 2005. Mercury levels in Bicknell’s thrush and other insectivorous passerine birds in montane forests of the northeastern United States and Canada. Ecotoxicology 14:223-240. (Abstract)
Rimmer, C.C. and K.P. McFarland. 2001. Known breeding and wintering sites of Bicknell’s Thrush. Wilson Bull.113: 234-236. (Abstract)
Hobson, K.A., K.P. McFarland, L.I. Wassenaar, C.C. Rimmer and J.E. Goetz. 2001. Linking breeding and wintering grounds of Bicknell’s Thrushes using stable isotope analyses of feathers. Auk 118:16-23. (Abstract)
Rimmer, C.C. and J.R. Tietz. 2001. An adult male Blackpoll Warbler in female-like plumage. J. Field Ornithol. 72:365-368. (Abstract)
Rimmer, C.C. and K.P. McFarland. 2000. Post breeding dispersal and migration stop-over in a montane spruce-fir forest in northern Vermont. Wilson Bull. 112:124-136. (Abstract)